Using Hydrogen Peroxide to Disinfect Cleanroom Surfaces
Using Hydrogen Peroxide for Cleanroom Disinfection
The control and removal of contaminants are essential to the proper operation of a cleanroom environment. To avoid the often catastrophic impact of contamination, cleanroom operators must adhere to strict processes and protocols. In a cleanroom, one of the most critical control activities involves the disinfection of all surfaces. The quality of regular surface disinfection wipedown procedures can make the difference between a high-quality, safe, problem-free cleanroom and a situation that can lead to risks to an organization's reputation, fines, regulatory trouble, and even consumer illness. There are several approaches to cleanroom disinfection, one of the most effective methods employs hydrogen peroxide.
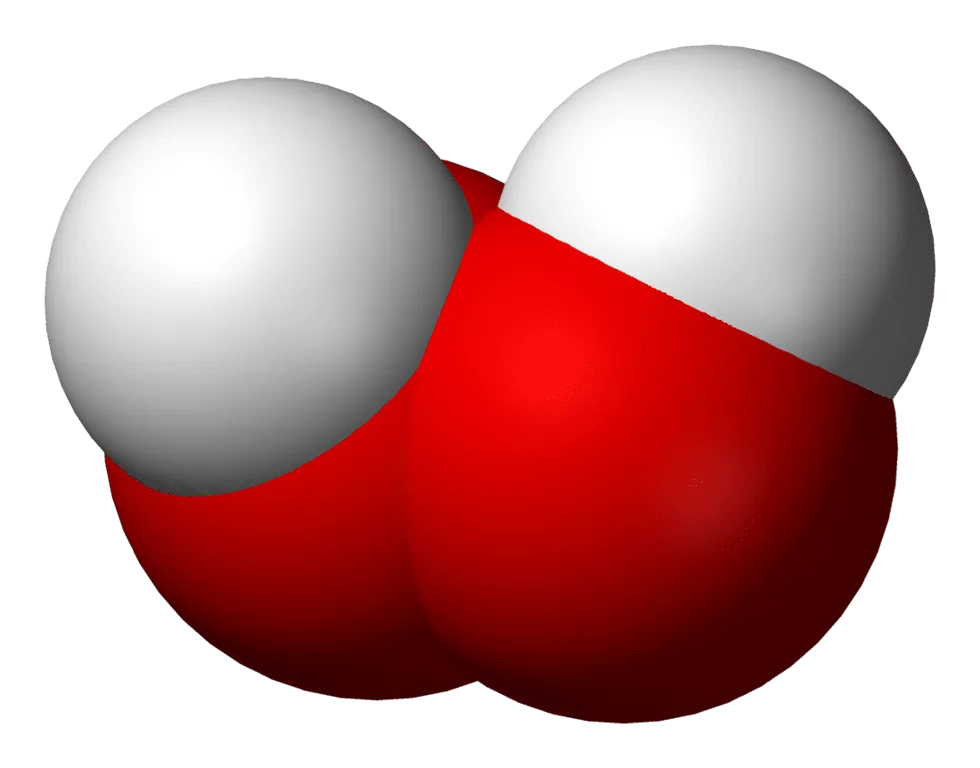
Hydrogen Peroxide - 3D
Space filling model of the hydrogen peroxide molecule
What Is Hydrogen Peroxide?
Most of us are familiar with hydrogen peroxide (H2O2), which is often kept on hand in a first aid kit for wound care. It is a quick way to destroy bacteria before bandaging small cuts. H2O2 is chemically simple. It is two oxygen atoms, each bonded to its own hydrogen atom and then bonded together. It is very similar to water except with an extra atom of oxygen, which gives it additional power to destroy bacteria and other microorganisms.
How Does Hydrogen Peroxide Work as a Disinfectant?
Hydrogen peroxide contains one additional atom of oxygen when compared to regular water. It is that atom that gives it its disinfecting properties. While the evidence of its effectiveness is clear, the process by which it works is still debated. It is believed that it works through the process of oxidation, with the compound's oxygen atoms stealing electrons from the cell walls of bacteria and causing them to break apart.
How is Hydrogen Peroxide Used in Disinfecting Cleanroom Surfaces?
Hydrogen Peroxide is typically diluted in water for a 3-6% solution. It is used in conjunction with cleanroom wipers, which feature lower levels of lint to avoid contamination and offer higher than normal levels of absorbency and chemical resistance. The solution can be applied directly to the wipers and then used immediately on hard surfaces. For convenience, pre-packed wipes already wet with hydrogen peroxide solution can be used.
Whichever method used, it is critical to be aware of the unstable nature of hydrogen peroxide. Over time, it breaks down into simple oxygen gas and water, reducing the concentration of H2O2 within the solution and reducing the effectiveness of disinfection.
For this reason, it is usually packaged in opaque bottles to slow the process by eliminating exposure to light. Prepackaged wipes may be vulnerable to shorter decomposition time due to the packaging, so it is essential to keep track of the age of inventory and have a rigid protocol in place for replacing outdated supplies.
Cleaning Protocols
When hydrogen peroxide is used as a disinfectant, it must be applied to a bare surface free of soil or any other debris. Anything left on the surface will consume the hydrogen peroxide and limit its disinfecting ability. Any necessary cleaning must be attended to before disinfection. Protocols must be developed and followed to ensure that any soil is removed using detergent. Then, before the application of hydrogen peroxide, the surface must be rinsed with deionized water.
Once surfaces are clear of soil and detergent, they should be wiped with the cleanroom wiper and hydrogen peroxide using linear wiping strokes, always moving from the cleanest to the dirtiest area to avoid recontamination. All surfaces should be allowed to air dry to allow the disinfection process to complete. There is no chemical residue to be concerned about with hydrogen peroxide. However, a second wipedown is recommended, again with hydrogen peroxide and cleanroom wipers, to remove any endotoxins left from the disinfection process.
Protocols Matter
Sanitation with hydrogen peroxide is proven effective for the disinfection of cleanroom surfaces—however, the solution itself is only one part of a clear process that must be followed precisely. A poorly mixed solution, out of date supplies, failure to remove soil, or other missteps can reduce or eliminate the disinfecting impact and leave bacteria behind. Whether you use hydrogen peroxide or another method, ensuring that all personnel follow procedures exactly will mean the difference between effective disinfection or the high cost of contaminants spreading within the cleanroom environment.
The information supplied in this article is for guidance only. Not all cleanrooms will use the same procedures. Follow your specific cleanroom or company procedural manual before this guide.