Endotoxin Control in Pharma and Medical Device Manufacturing
What is an endotoxin?
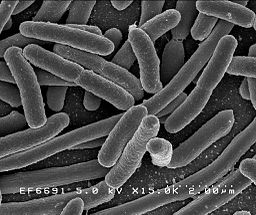
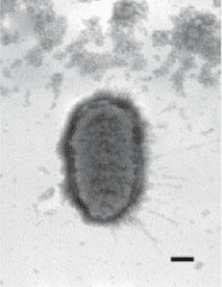
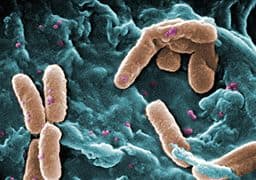
An endotoxin is a lipopolysaccharide (LPS) found in the cell wall of gram negative bacteria1.
The term "microbial pyrogen" as opposed to "gram negative bacterial endotoxin" has been used to describe many different substances. It should be noted that pyrogenic substances can be produced by some gram-positive bacteria, mycobacteria, fungi and also viruses, but the pyrogens produced by gram negative bacteria, i.e., the endotoxins, are of significance to the pharmaceutical and medical device industries.
Located in the outer membrane of gram-negative bacteria, Bacterial endotoxins fall within a class of phospholipids called lipopolysaccharides (LPS). LPS are not exogenous products of gram negative bacteria. The release of LPS from bacteria takes place after death and lysis of the cell. Examples of pyrogen producing gram-negative bacteria are E-coli, Proteus, Pseudomonas, Enterobacter and Klebsiella.2
Pyrogens and more specifically, endotoxins, pose a significant safety concern if a product becomes contaminated. For this reason, the prevention of endotoxins in the manufacturing process is critical.
Endotoxin Limits for Individual Pharmaceutical Products
According to the FDA, the formula used to calculate endotoxin limits for drug products varies based on the dose that is administered to the patient. Because of this, the endotoxin limit is expressed as K/M.
K is 5.0 EU/kilogram (kg.), which represents the approximate threshold pyrogen dose for humans and rabbits. That is the level at which a product is adjudged pyrogenic or non-pyrogenic. M represents the rabbit pyrogen test dose or the maximum human dose per kilogram that would be administered in a single one hour period, whichever is larger. If a product is labeled for intrathecal injection, then K is 0.2 EU/kg. However, there are 5 water products (previously discussed) which, because of the large volumes that can be administered and absence of dose limitation, have specific endotoxin limits per ml.
Endotoxins Limits for Medical Devices
The Center for Devices and Radiological Health (CDRH) implemented the USP Endotoxin Reference Standard and limits for medical device extracts expressed in EU/mL.
The endotoxins limit for a medical device is dependent on the intended use of the device and what the device contacts (e.g., blood, the cardiovascular system, cerebrospinal fluid, intrathecal routes of administration, permanently implanted devices, and devices implanted subcutaneously).3
Preparing a product for evaluation varies from device to device. Some medical devices can be flushed, some may have to be immersed, while others may need disassembly. Refer to USP Chapter 161 Transfusion and Infusion Assemblies and Similar Medical Devices for guidance and limits on medical devices.
Products for Endotoxin Contamination Control
Using the appropriate products in the manufacturing and cleaning process can help minimize the risk of endotoxin contamination. Products such as IPA, Hydrogen Peroxide, and Cleanroom Wipes can be tested for endotoxin levels.
Please contact us for more information about products for endotoxin control.
Sources:
- What is Bacterial endotoxin https://labchem-wako.fujifilm.com/asia/lal/lal_knowledge/about_lal.html ↩
- ITG SUBJECT: BACTERIAL ENDOTOXINS/PYROGENS https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-technical-guides/bacterial-endotoxinspyrogens ↩
- Guidance for Industry Pyrogen and Endotoxins Testing: Questions and Answers https://www.fda.gov/media/83477/download ↩